Narcissism has become a popular modern buzzword, but the dilution of its meaning from its mythological context and its misuse in clinical definitions has contributed to a lack of appreciation for the dire social and personal consequences of tolerating narcissistic abuse.
In modern times, when we think of narcissism we tend to point to “selfie culture” and a saturation of self-centeredness among those who enjoy it. Just yesterday I had the perfect opportunity to witness this while at a restaurant for lunch. A middle-aged woman with dyed black hair and matching bright red lips and nails had just entered her car, which was directly opposite the window where I was seated.

I watched, laughing to myself, as she spent the next five minutes taking pictures of herself in different poses: pouting, smiling, flipping her hair around, like she was a sexy actress on stage instead of a no-longer-attractive woman in a huge black SUV, just pretending. I imagine she has all the latest filters on her camera’s lens and wherever she’s headed next will be dark enough that the illusion is not prematurely disolved.
And while this is really cringy, its not particularly dangerous for anyone else but the poor sap she gets her hooks in next. Sorry dude, better luck next time!
Clinically, and even historically, this is not really the brand of narcissism that is causing so much mayhem in our world.
However, clinically the definitions are at once well-defined, and nebulous, and over-lapping, and found under the heading of the ‘dark triad’ personality disorders. While these are certainly interesting to study and are attempting to become more scientific with more data over time, they aren’t necessary to understanding why these personality disorders become so socially destructive.
The problem is not the cringy aging woman who wants to maintain the illusion of youth and beauty indefinitely in order to gain sexual approval in the marketplace. The problem is when the fantasy is allowed to trump the reality.
Society does still associate selfie-culture with the myth of narcississus, but only superficially. We correctly identify the beautiful youth who falls in love with his image in the water as narcississtic. But, we fail to look deeper into the lesson the ancient myth is attempting to teach us.

While there is no one official narcissus myth, one popular version includes his female companion, Echo. Whereas Narcissus is the disembodied face, Echo is the voice which mimics his, repeating, without agency, but symbolizing the intention of love and care through her adoration of him.
The real lesson is not just about the superficiality of beauty that fades, or the personal destructiveness of being overly-fascinated by one’s own appearance or superficial desires, but about the inherent dangers of illusion, especially when those illusions echo through the culture, without agency, repeating the words of the narcissist.
Reach out to touch the beautiful illusion in the water and it dissappears, and with it so does its echo, that’s the essence of the myth. There is no substance there, it’s a mirage. The face has no body, no core, no real form.
In popular culture today, Echo’s role represents the ‘flying monkeys,’ which are those figures doing the bidding of the narcissists, spreading his delusions. Narcissists are experts at manipulating others into doing their dirty work, whether that be their dishes or their crimes.

In psychoanalytical terms, this means the narcissistic personality has not done the difficult work of developing an authentic self. There is nothing behind the mask. S/he is a shape-shifter, a cameleon, the disembodied creature in a shared fantasy.
When these attributes become socially acceptable, indeed institutionalized, the culture itself becomes permeated with such types. Toxicity becomes the norm. Fantasy, being the preferred state of the masses, replaces reality. Narcississtic abuse becomes systemic.
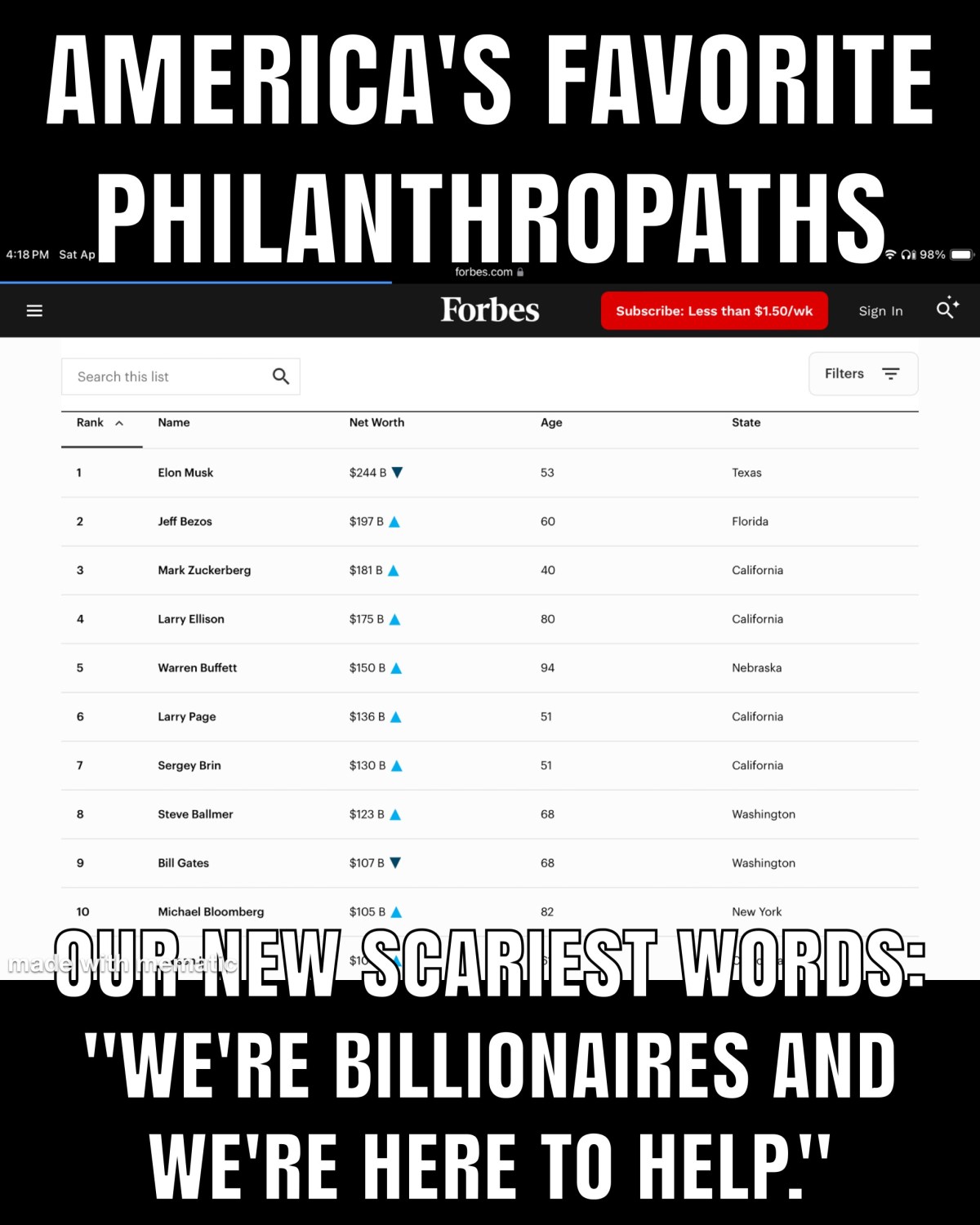
At the societal level, Geoengineering presents a typical example of how this looks in action. And, there are a great many direct parallels between narcississtic abuse as it plays out in personal relationships and in the aggregate, that is, the public domain.
Empty promises like ‘safe and effective’ become accepted as truth. Meaningless words and slogans replace honest dialogue and debate. Hollow gestures replace accountability.
In the personal relationship this looks like the philandering spouse who swears they’ve cheated for the last time, over and over. Or the sister or friend who apologizes for wrongdoing, but then hides behind ignorance or innocence, or tries to blame shift and gaslight to get out of being held accountable for her poor actions or insensitive behavior.
In the public sphere this normalized behavior becomes the governments and institutions who are granted immunity, the banks that are too big to fail, and the laws that are twisted to absolve the guilty and victimize the innocent.


What have we been told for the last decades about Geoengineering? At first we were told not to believe our lying eyes, nothing is happening, ‘chemtrails’ aren’t real, and you are all gullible conspiracy-believing nutjobs.

Next, we are told that it’s actually benevolent, they are trying to fix the broken climate. Totally safe and effective. We just need the states to put a few crucial laws in place, then everyone will be happy. Fantasies and illusions replace accountability.
More empty promises, more hollow gestures — like the states stalling with laws that will never stop the assault, but will get a new market brewing for all the attorneys and advocacy groups and health care professionals, who will make a lot of money from the fallout. Entrepreneurship, that will solve it. More tech, better tech, that will solve it.
Accountability is for ninnies.
“A government entity, its officers, and its employees, are immune from suit, and immunity is not waived, for any injury proximately caused by a negligent act . . .”
Utah https://zerogeoengineering.com/2026/utah-house-bill-79-provides-governmental-immunity-amendments/House Bill 79 Provides Governmental Immunity Amendments – Zero Geoengineering
The argument gets twisted around intentionality, did they mean to harm, or not? Let me go out on a huge limb here and suggest that every single person involved will say they didn’t intend to harm. That’s rocket science.

That question itself serves the abusers. The real question, the one the victim would ask is, “Should abusers be able to hide behind innocence, ignorance or incompetence?”
Solar Geoengineering and the Global Commons—What Role for Ecocide Law? | Springer Nature Link
“As a result, initially peaceful, yet ultimately harmful geoengineering projects could exemplify unintentional hostile use of weather modification technologies. However, ENMOD does not take this form of use into account, so cannot provide a viable tool.”
Meaningless words–words that magically transform, or lose and gain significance depending on whims and propaganda shifts–global warming, global cooling, climate change, abrupt climate shift, climate remediation.
These parallel the meaningless words and hollow gestures of the narcissitic abuser — I’m sorry, I didn’t meant to, it’s really not that bad, it’s just your impression. The dog ate my homework.
It’s not my fault. No agency. No accoutability.
You’re not perceiving what you think you are perceiving! You’re not feeling what you think you’re feeling! it’s just your impression! Your subjective experience! If it were true, everyone would be experiencing it exactly the same like you!
Plausible deniability is the safe space for abusers of all kinds.
They will even go as far as admit the wrongdoing, but still not be held accountable for it. Like Narcissus, all image with no substance, all face with no body.
Other common tricks of the trade: manipulation of perception, moving the goal post, feigning, finessing, presenting harmful acts as benevolent–it’s for your own good, for the good of the community, because they are so concerned about you, about the environment.
What do you mean we can’t flood your home? But the farmers need rain, you’re being selfish. Isn’t that why you bought insurance? Why aren’t you more resilient? Why should you be so upskittled by a tornado or two, they happen all the time. We were just doing our job!
I will NOT end this post, or this life, drowning in their sea of dysfunctions, because that’s exactly what works best, FOR THEM.
I truly believe that if the individual will stop accepting such abuses in their personal lives, what’s acceptable on the societal level will assuredly, eventually, shift as well, and very dramatically.
Stop approving of the toxic behavior, stop believing the lies, stop being their Echo, because narcissistic abusers are pathological attention-seekers. Call them out, demand accountability, do not accept empty promises and meaningless words, and watch as the little rats start to scurry.
Learn about narcississtic abuse and root it out of your life, personally and professionally. Achieve greater levels of discernment through diligence and determination. Real resilience comes from principled positions rigorously and consistently applied. Insist others practice what they preach, as you yourself do the same.
This is not idealism, this is the tough work of forcing reality to win over illusion. It is the tough work of embodiment, of authentic Self creation.
“We’re often told that narcissism doesn’t exist or that we’ve attracted these people into our lives because of our own issues. This harmful victim-shaming keeps people trapped in abusive dynamics.
In this episode, Dr Peter Salerno explains that narcissism is real and identifiable. Narcissists are invested in their image at the expense of their true self, and they intentionally seek out environments where they can exploit others.
This conversation will help you identify if you’re dealing with a narcissist, trust your body’s reaction to mistreatment, and begin to think about whether it’s a relationship you want to remain in.”
We may still be victims of their floods and tornadoes and varied toxic tampering, but we don’t have to accept their delusional fantasies and their illusions of control. And that WILL make a difference.
Eventually.